Introduction: Specific assay of plasma rivaroxaban level are not always readily available with short turn-around-time which hampered the management of urgent clinical situations. The response of routine coagulation screening tests show huge lot-to-lot differences with the plasma rivaroxaban level. International normalized ratio (INR) is a standardized measurement taking into account of lot-to-lot variations of PT reagents. In this study, we aimed to build a predictive formula of plasma rivaroxaban levels from INR value and validated in real world clinical situations.
Method: The study period was 1 st January 2018 to 31 st December 2021. Ninety-four patients who taking rivaroxaban participated in the study. Patients were randomized into testing cohort and validation cohorts. The prediction formula was built from the testing cohort and then validated in validation cohort. The predictive performance was further validated on real-world clinical requests.
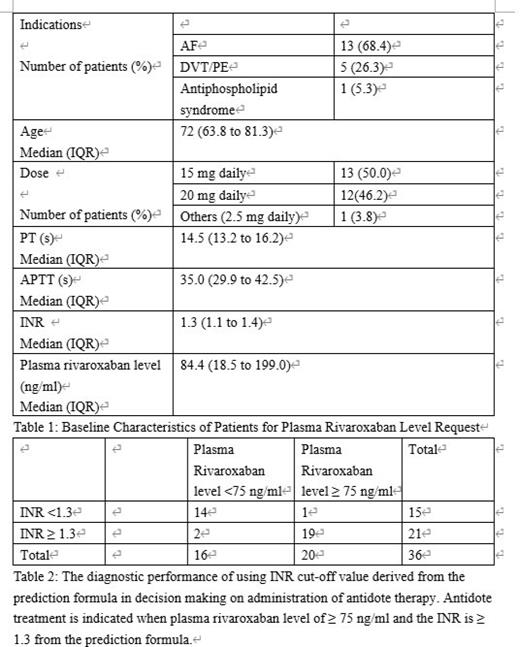
Results: The baseline characteristics of both cohorts were well-balance. There was significant correlation between the plasma rivaroxaban level and PT in both the testing and validation cohorts (Testing cohort: R square 0.838, p < 0.01; Validation cohort: R square 0.836, p < 0.01). The plasma rivaroxaban level showed significant correlation with APTT in both the testing and validation cohorts (Testing cohort: R square 0.744, p < 0.01; Validation cohort: R square 0.654, p < 0.01). There was also significant correlation between INR and plasma rivaroxaban levels in both the testing and validation cohorts (Testing cohort: R square 0.819, p <0.01; Validation cohort: R square 0.816, p < 0.01). Both INR and plasma rivaroxaban levels were undergone natural logarithmic transformation. We choose the quadratic model to determine the relationship between the natural log of plasma rivaroxaban level and the natural log of INR as it had highest R square value (R square 0.850, p<0.01). The formula is: ln (plasma rivaroxaban level) = 2.96+7.87 X ln (INR) - 5.04X (ln (INR))^2. The root mean square error (RMSE) of the predictive formula for the testing and validation cohorts were 61.81 ng/ml and 69.32 ng/ml, respectively. We received a total of 43 requests for plasma rivaroxaban assay involved 29 patients during the study period (Table 1). Three patients had multiple requests for plasma DOAC level assays during this period. The cut-off INR value was 1.3 for administration of antidote for rivaroxaban, which corresponded to a rivaroxaban level of 75 ng/ml. The sensitivity and specificity for using this INR cut-off value to determine the threshold plasma rivaroxaban level (75 ng/ml) for administration of reversal agents were 95% (95%CI: 85.4 -100%) and 87.5% (95%CI:71.3-100%) respectively in real world clinical situations (Table 2).
Conclusion: Plasma rivaroxaban level can be calculated from prediction formula by INR value with satisfactory accuracy and the INR cut-off value derived from it can be used to guide the decision for administration of reversal agents with excellent sensitivity and specificity.
Disclosures
No relevant conflicts of interest to declare.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal